|
3/9/2024 0 Comments Hydrogen atom bohr model
The Neils Bohr atomic version speculation considers electrons to have each recognised function and momentum simultaneously, that’s unthinkable as indicated with the aid of using Heisenberg. Bohr’s model no longer observes the Heisenberg Uncertainty Principle. The study recommends that current quantum atomic models and equations governing quantum physics should be revisited for better applications and understanding of the physical world. The following are the fundamental limitations of Bohr’s Model of the hydrogen atom. For a deeper understanding, these constants may be scrutinised for their determination. They can also be derived from the steady state hydrogen atom Schrodinger's equation. The Bohr model of the hydrogen atom (Z 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (h). The four fundamental physical constants can be applied to multi-electron systems because they are inherent characteristics of an electron. There are two other constants \(\delta\) = 7.199822 × 10 -10 eVm and \(\zeta\) = 1503796277m -1 that were identified in the Bohr’s equation of the hydrogen atom. Bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. Main Idea A visualization of the Bohr model and the hydrogen spectrum. Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. In 1911, Rutherfords - particle experiments were very controversial The idea that all the positive charge of an atom was crammed into the nucleus was hard for many to accept. 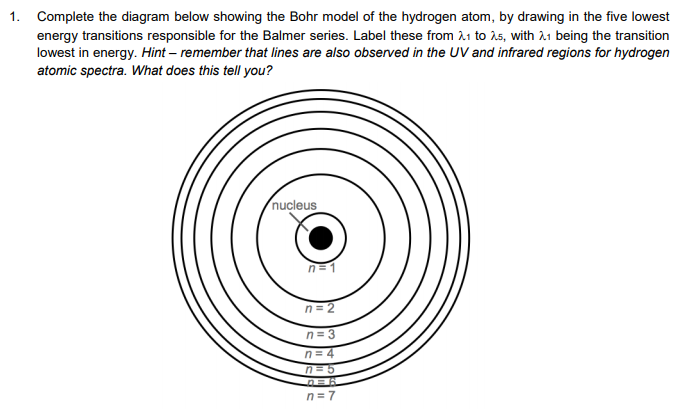
The aim for the review was to contribute to the solution of disagreement between the Bohr’s wavelength (\(\lambda\) 0 = 91.12nm) and the Balmer’s experimental observation (\(\lambda\) 0 = 91.18nm) for the emission spectrum of hydrogen atom. Bohrs model of the hydrogen atom was the first to incorporate quantum theory, and the key idea of his model was that electrons occupy discrete orbitals. Light, he proposed, radiated from hydrogen atoms only when an electron made a transition from an outer orbit to one closer to the nucleus. U(r) ke2 r, where k 1 / 40 and r is the distance between the electron and the proton. In the electric field of the proton, the potential energy of the electron is. With the assumption of a fixed proton, we focus on the motion of the electron. By limiting the orbiting electrons to a series of circular orbits having discrete radii, Bohr could account for the series of discrete wavelengths in the emission spectrum of hydrogen. Figure 8.2.1: A representation of the Bohr model of the hydrogen atom. Two values of fundamental physical properties of an electron in the hydrogen atom has been identified. This chapter has provided an alternate method of solving simple problems in atomic physics under Bohr’s model to aid good mental picture of hydrogen atom to scientists.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
